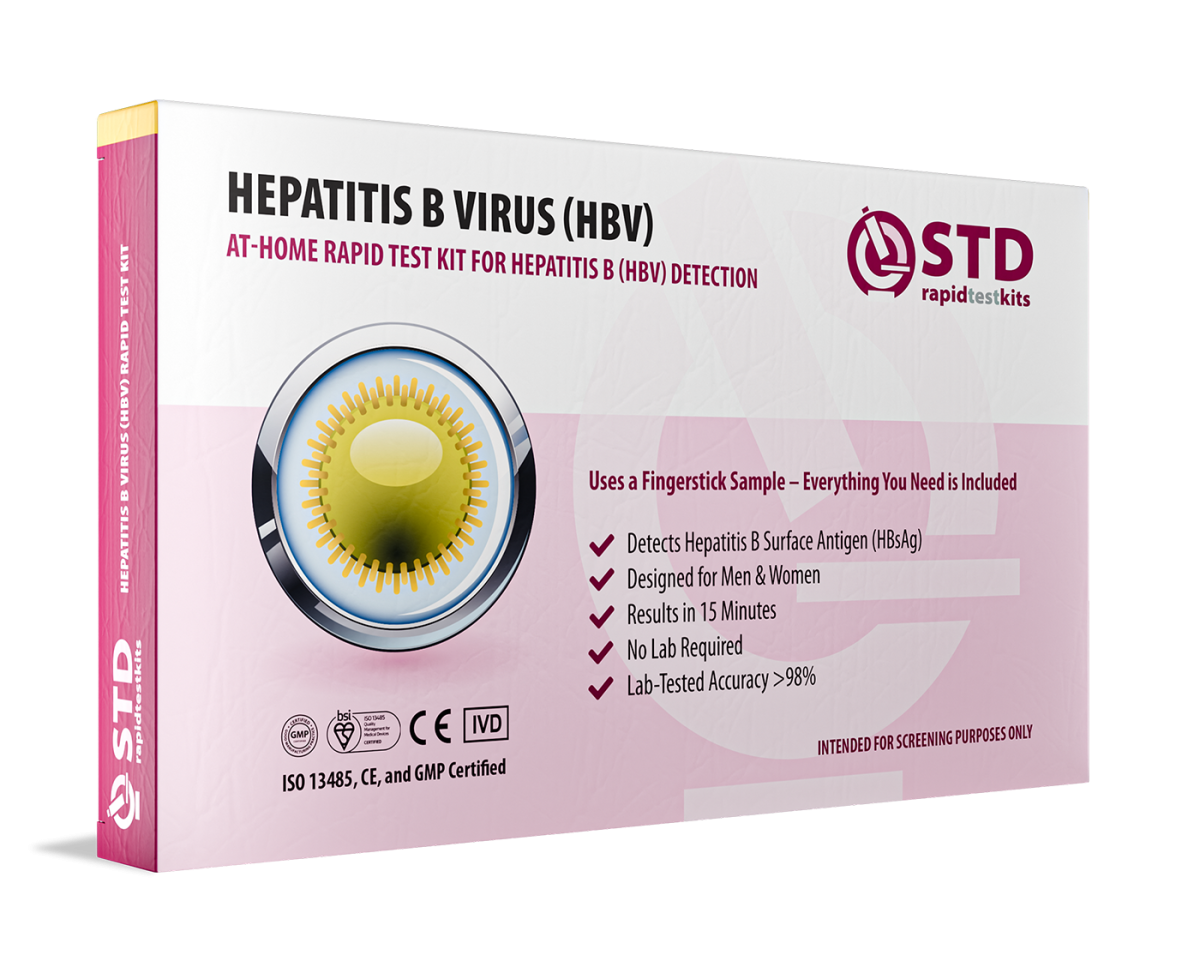
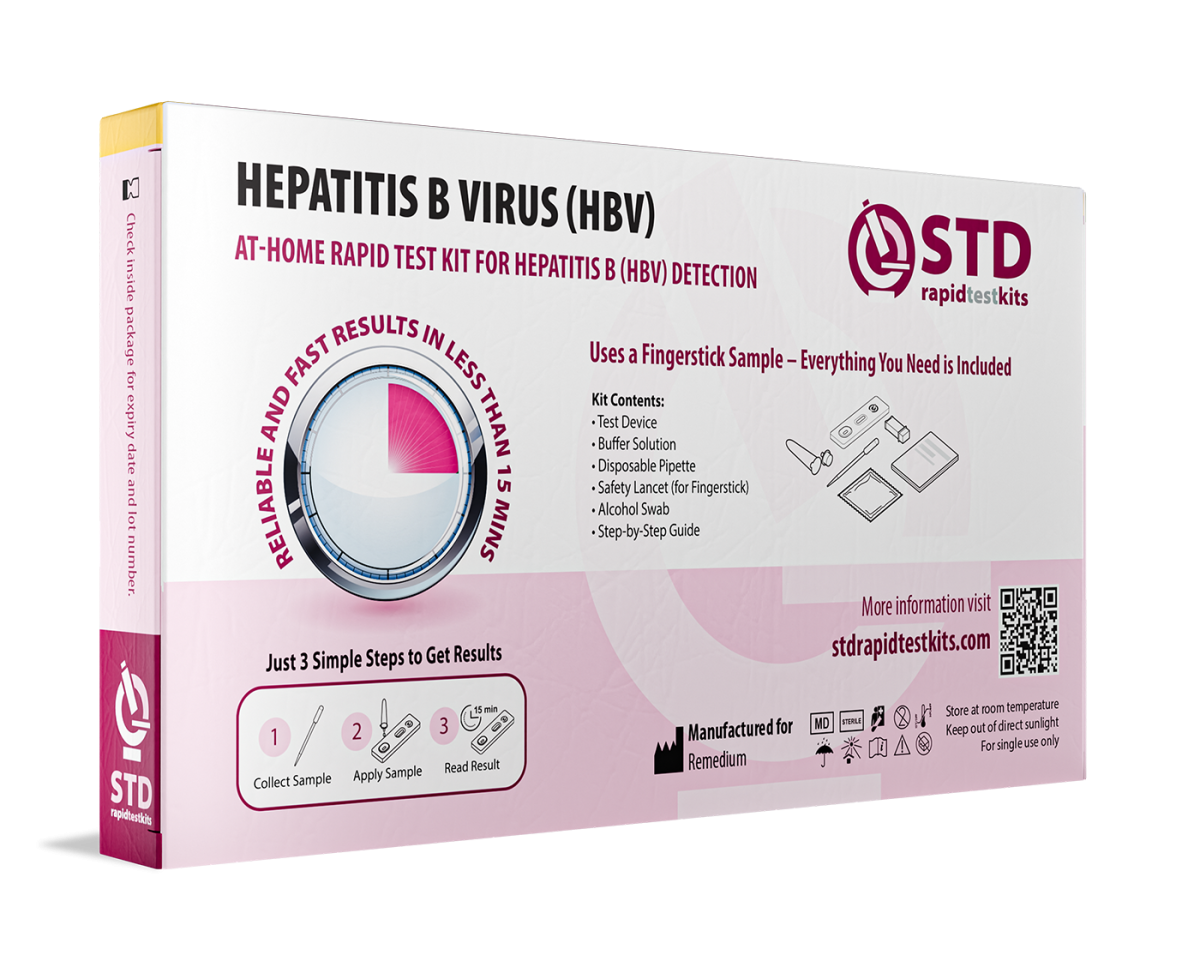
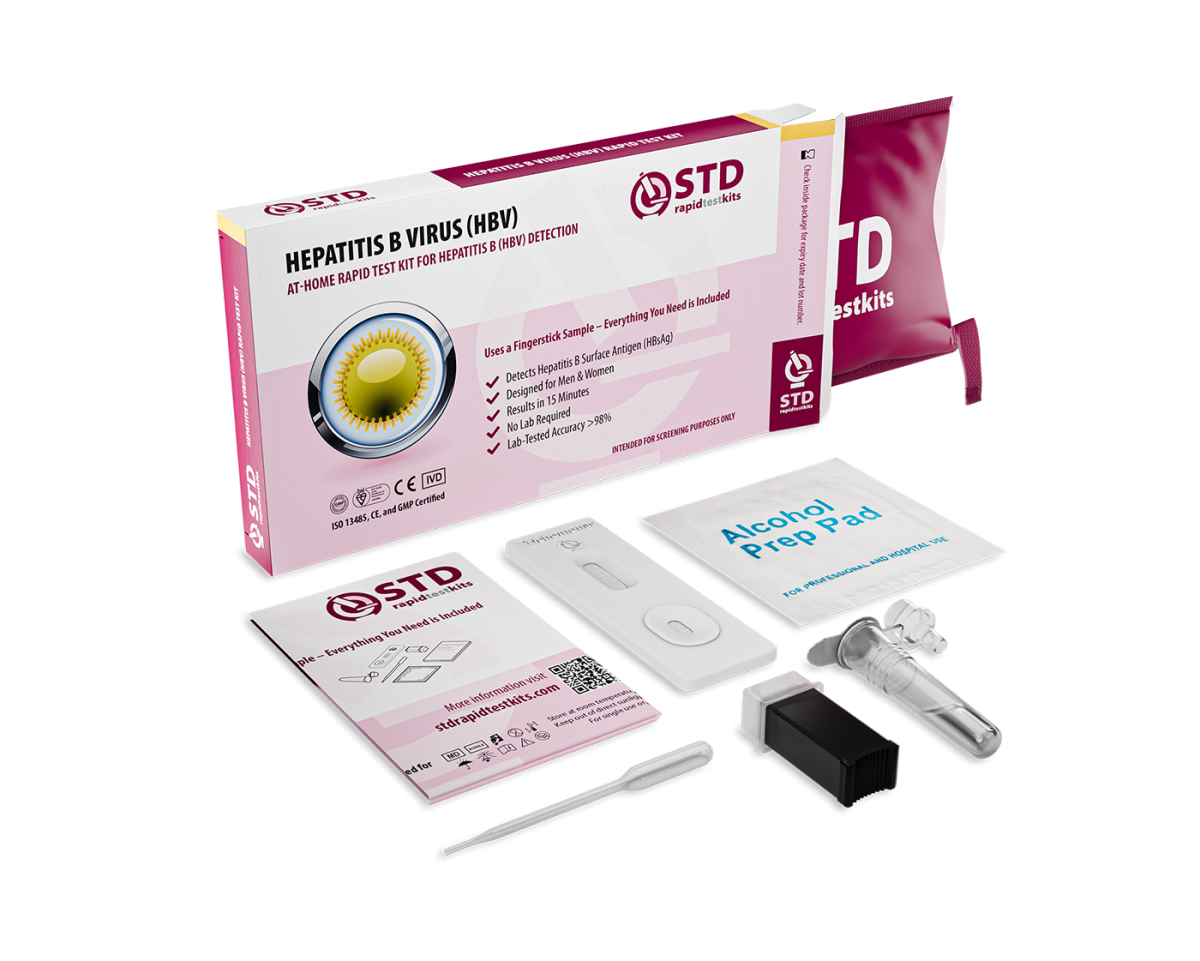
Hepatitis B is a contagious liver infection that often shows no early symptoms, making regular testing essential. Our Hepatitis B At-Home Rapid Self-Test Kit provides 98.8% accurate results in minutes, detecting Hepatitis B surface antigen (HBsAg) from a simple blood sample. The test is easy to use, requires no lab processing, and can be done privately at home.
Many people with Hepatitis B remain unaware of their infection, unknowingly spreading the virus. When symptoms do appear, they may include fatigue, jaundice, dark urine, nausea, abdominal pain, and joint pain. If left undiagnosed, Hepatitis B can lead to chronic liver disease, cirrhosis, liver failure, and liver cancer.
For full assurance, test once after potential exposure and retest after the 1 to 6-month incubation period to confirm your results. Since Hepatitis B and Hepatitis C may present with similar symptoms, consider adding a Hepatitis C test kit for comprehensive screening.